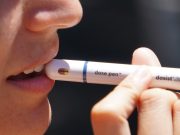
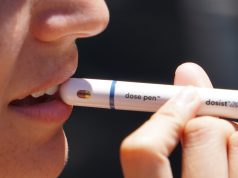
A constant battle against opioids in the U.S. is requiring experts every day to find ways to save lives. To confront the epidemic, two advisory committees from the federal Food and Drug Administration voted in favor of developing a nasal spray version of naloxone or Narcan that can be accessible to the public over the counter.
The life-saving drug works by blocking opioid brain effects for approximately 90 minutes and helping to restore breathing.
According to a recent report from the CDC, the number of opioid overdoses in the U.S. rose by 17% in 2021. Thirty-five people of every 100,000 died from an overdose last year in Florida.
“It’s this thing that you want to have in your home for safety and you hope you never would have to use it”, says an FDA representative.
The FDA is working on label instructions for easier use. Once approved, the overdose antidote could be found in grocery stores, gas stations, and bending machines across the country.